PUREGIA Technology
Fresh Hypochlorous Acid Generated On Demand
PUREGIA is a patented technology platform designed to generate fresh hypochlorous acid disinfectant solutions on demand through controlled electrochemical activation.
This system combines solid tablet chemistry with electrolysis to produce highly effective disinfecting solutions while maintaining stability during storage and transport.
PUREGIA Technology Overview
PUREGIA is a patented system designed to generate fresh hypochlorous acid (HOCl) disinfectant solution on demand through a combination of solid tablet chemistry and controlled electrolysis.
Hypochlorous acid is widely recognized as one of the most effective disinfectants against bacteria, viruses, and other microorganisms. However, traditional HOCl solutions are difficult to store because the active chlorine gradually decomposes over time.
The PUREGIA system addresses this challenge by separating long-term storage from disinfectant generation.
Instead of storing hypochlorous acid directly, PUREGIA stores carefully balanced components in a stable solid tablet form. When the tablet is dissolved in water, it prepares the proper electrolyte solution required for electrolysis.
Hypochlorous acid is then generated only when electrical current is applied, producing fresh disinfectant solution at the moment it is needed.
This approach allows PUREGIA to combine the stability of solid materials with the effectiveness of freshly generated hypochlorous acid, providing a practical and reliable method for disinfectant production.
Why Hypochlorous Acid Has Not Been Widely Used
Hypochlorous acid (HOCl) is widely recognized as one of the most effective disinfectants against bacteria, viruses, and other microorganisms. It acts rapidly, leaves minimal residue, and is generally safer for human contact when used at appropriate concentrations.
Despite these advantages, hypochlorous acid has historically not been widely adopted for general household use. The main reason is that HOCl solutions are difficult to produce, chemically unstable, and challenging to store for extended periods once prepared.
Because of these limitations, most disinfectants commonly used today rely instead on sodium hypochlorite (bleach) or other chlorine-based compounds.
Conventional Methods of Producing Hypochlorous Acid
Sodium Dichloroisocyanurate (NaDCC)
One commonly used method for generating disinfecting chlorine solutions is by dissolving Sodium Dichloroisocyanurate (NaDCC) tablets or powder in water.
When NaDCC dissolves, it produces available free chlorine (AFC) measured in parts per million (ppm). Through chemical equilibrium reactions, a portion of this chlorine exists as hypochlorous acid (HOCl), the active disinfecting agent.
This approach has been widely used for swimming pool sanitation. However, it has not been commonly adopted for household use because NaDCC products typically contain very high chlorine concentrations, making accurate dilution and safe handling difficult for ordinary users.
For example:
Target Concentration NaDCC Required. Water Required
50 ppm 0.5 g 6 L
100 ppm 0.5 g 3 L
200 ppm 0.5 g 1.5 L



Electrolysis of Salt Water
Another method used to generate disinfecting chlorine solutions is electrolysis of salt water.
In this system, water is mixed with salt (sodium chloride) and sometimes a pH reducer, such as vinegar. When electrical current is applied, chloride ions in the solution are converted into chlorine-based disinfecting compounds.
However, without proper control of the solution chemistry, electrolysis may produce sodium hypochlorite (bleach)instead of hypochlorous acid.
The PUREGIA Method
The PUREGIA system was developed to combine the stability of solid formulations with the ability to generate fresh hypochlorous acid on demand through electrolysis.
PUREGIA utilizes a specially engineered solid tablet designed to prepare the correct electrolyte solution before electrolysis occurs.
When the tablet is placed in water, sodium bicarbonate reacts with sodium diacetate, producing carbon dioxide gas (CO₂). This effervescent reaction rapidly breaks apart the tablet, allowing the ingredients to dissolve quickly and disperse evenly throughout the water.
At this stage, the solution does not yet contain hypochlorous acid. Instead, it forms a prepared electrolyte solutioncontaining dissolved salt and a carefully regulated pH environment.
When this solution is subjected to electrolysis, chloride ions are converted into active chlorine species. Because the chemical conditions have been carefully balanced, the chlorine produced exists primarily as hypochlorous acid (HOCl)rather than sodium hypochlorite.
This approach allows PUREGIA to generate fresh hypochlorous acid only when needed.
What Is Inside a PUREGIA Tablet?
Each PUREGIA tablet contains a carefully balanced combination of components that prepare the solution for electrolysis.
Salt (Sodium Chloride)
Provides the chloride ions required for electrolysis.
Sodium Diacetate
Acts as a controlled pH regulator, helping maintain the optimal chemical environment for hypochlorous acid generation.
Sodium Bicarbonate
Reacts with sodium diacetate when dissolved in water, producing carbon dioxide gas that rapidly breaks apart the tablet and accelerates dissolution.
Together, these ingredients create a prepared electrolyte solution, from which hypochlorous acid is generated through electrolysis.
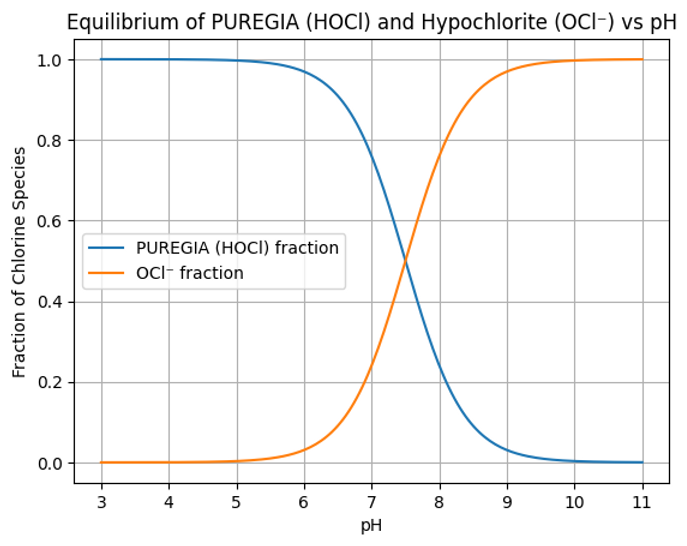
Equilibrium of PUREGIA (HOCl) and Hypochlorite (OCl⁻) vs pH
The disinfecting chlorine species in water exist mainly as hypochlorous acid (HOCl) or hypochlorite ion (OCl⁻). The relative proportion depends strongly on the pH of the solution. Under properly controlled conditions, the PUREGIA system favors the formation of hypochlorous acid, the more effective antimicrobial species.
Stability of PUREGIA Hypochlorous Acid Solution
One challenge associated with hypochlorous acid solutions is their limited stability after production.
Testing of the PUREGIA system shows that when the generated solution is stored in a sealed PET bottle at room temperature, the hypochlorous acid concentration decreases gradually over time.
Under typical indoor storage conditions, approximately 50% of the initial hypochlorous acid concentration remains after about seven weeks (49 days).
Because hypochlorous acid naturally decomposes over time, users are encouraged to prepare fresh solution periodicallyto ensure optimal disinfecting performance.
The natural decay of hypochlorous acid also contributes to environmental safety, as the solution gradually reverts to ordinary saline.

International Patent Protection
The technology behind the PUREGIA system is protected by an international patent portfolio covering the formulation and process used to generate hypochlorous acid through controlled electrolysis.
Patent protection has been obtained in the following jurisdictions:
Japan — Patent No. 6986797
Granted December 2, 2021
China — Patent No. 6685511
Granted February 6, 2024
Hong Kong — Patent No. 6986797
Granted April 19, 2024
United States — Patent allowed by the USPTO, issuance pending
The PUREGIA technology platform is currently in the early stages of commercial development.
